 Certificates of analysis are necessary to inform customers about important characteristics of purchased reference standards such as concentration of the toxin(s), date of expiration, and storage recommendations. Without a certificate of analysis, this vital information would not be as easily accessible to the customer. These certificates contain all pertinent information about the given standard, all in one place.
Certificates of analysis are necessary to inform customers about important characteristics of purchased reference standards such as concentration of the toxin(s), date of expiration, and storage recommendations. Without a certificate of analysis, this vital information would not be as easily accessible to the customer. These certificates contain all pertinent information about the given standard, all in one place.
Every reference standard we sell comes with a certificate of analysis. Trilogy offers both stock and custom reference standards. Below is an example of a Trilogy certificate of analysis along with explanations for each section.
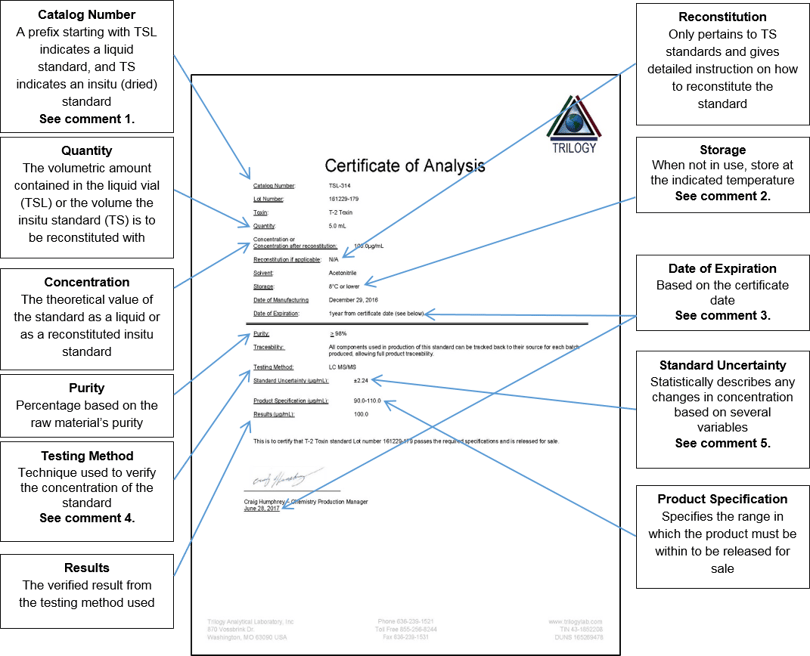
Comment 1: Prefix TSL identifies the standard as a liquid standard; TS identifies standards that have been dried and need to be reconstituted to the quantity volume listed on the certificate. Some part numbers may contain additional information. For example, for TSL-314-10 the TSL indicates that it is a liquid standard, the 314 indicates that it is a 100µg/mL T-2 standard in acetonitrile, and the 10 means that it is packaged in a 10mL quantity. This is useful when ordering custom volumes other than the stock volumes that we sell.
Comment 2: Standards should be stored at 8°C or lower to ensure concentration and stability of the standard when not in use. Standards should be removed from storage and allowed to come to room temperature before use. Once at room temperature, they should be vortexed (mixed) for 10 seconds before use. After using, if not needed for the rest of the day, they should be returned back to the listed storage conditions.
Comment 3: We have performed in house real time stability studies under the following storage conditions: freezer (-10 to -20°C), refrigerator (3-10°C), and room temperature (20-25°C) for 3 years. TSL standards are stable for 1 year from certificate date and TS standards are stable for 12 months before reconstitution and 6 months after reconstitution. The stability data applies only when stored in the original packaging at conditions listed on the certificate.
Comment 4: We use several different techniques to verify the concentration of the standards we produce. Depending on the toxin or if it is a mix of toxins, we may use one or more of the following techniques to verify the concentration: spectrophotometer using published extinction coefficients, HPLC, UHPLC, LC MS/MS or GC. When testing using HPLC, UHPLC, LC MS/MS or GC, we compare the new standard to a previously approved lot of standard to verify the concentration.
Comment 5: We use the Guide to the Expression of Uncertainty in Measurement (GUM). The expanded uncertainty is equal to the combined uncertainty multiplied by a factor of 2 to give 95% confidence in the measurement of uncertainty. Calculations of uncertainty (k = 2) are based on the criteria outlined in the ISO guide to the expression of uncertainty in measurement (GUM) and take into account the following variables: purity uncertainty, volumetric devices uncertainty, combined analytical instrument uncertainty and technician pipetting repeatability.
Learn more about our stock and custom reference standards or see more examples of our certificates of analysis.

Share on Social Media: