 Mycotoxins are secondary metabolites produced by molds which can have significant negative effects on the health, productivity and profitability of livestock. Molds can grow and mycotoxins can be produced pre-harvest or during storage, transport, processing, or feeding. Mold growth and mycotoxin production are related to plant stress caused by weather extremes, insect damage, inadequate storage practices, and faulty feeding conditions. Environmental conditions such as heat, moisture, and insect damage cause plant stress and predispose plants in the field to mycotoxin contamination.
Mycotoxins are secondary metabolites produced by molds which can have significant negative effects on the health, productivity and profitability of livestock. Molds can grow and mycotoxins can be produced pre-harvest or during storage, transport, processing, or feeding. Mold growth and mycotoxin production are related to plant stress caused by weather extremes, insect damage, inadequate storage practices, and faulty feeding conditions. Environmental conditions such as heat, moisture, and insect damage cause plant stress and predispose plants in the field to mycotoxin contamination.
The first step in reducing the risk of mycotoxin contamination in livestock feed is to prevent mold growth in the field, during harvest, and during storage. If unacceptably high levels of mycotoxins occur, removal or dilution of the contaminated feed is preferable; however, it is sometimes impossible to replace contaminated feeds. The addition of mycotoxin binders to contaminated feed has proven to be a very promising approach in reducing the effects of mycotoxins. The theory is that the adsorbent binds the mycotoxins in the feed strongly enough to prevent the adsorption across the digestive tract of the consuming animal. Potential adsorbent materials include activated charcoal, aluminosilicates (clay, bentonite, montmorillonite, zeolite, phyllosilicates, etc.), complex indigestible carbohydrates (cellulose, polysaccharides in the cell walls of yeast and bacteria), and synthetic polymers such as cholestyramine and polyvinylpyrrolidone and derivatives.
There are many in vitro methods for analysis of mycotoxin adsorbents, which include detection by thin layer chromatography (TLC), high-performance liquid chromatography (HPLC), and enzyme-linked immunosorbent assay (ELISA). TLC is the least sensitive and 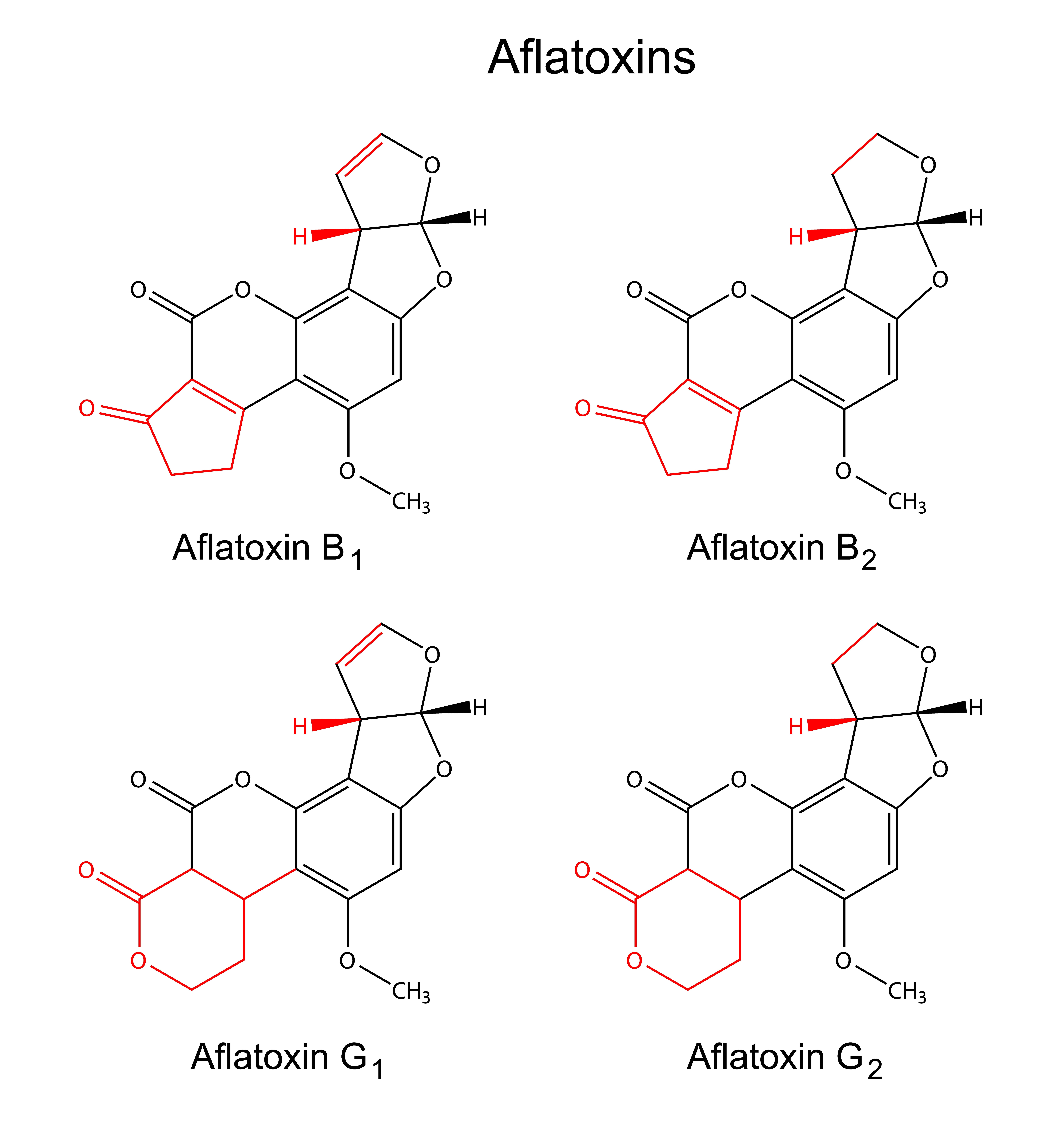 accurate of these methods. There are positives and negatives to both HPLC and ELISA methods. ELISA kits are easier to use and more cost effective, but they are more prone to matrix effects than HPLC analysis, which can cause false results. Another key limitation to ELISA kits is that they can only reliably quantify in a certain range, e.g. 0.3-10 ppb. Therefore, anything over the limit of the kit (in this example 10 ppb), needs dilutions in order to be accurately calculated, causing additional uncertainty in the results. HPLC does not have this limitation; results can be seen well outside of this range and accurately quantified. Another positive aspect of HPLC is the ability to distinguish between mycotoxins such as aflatoxin B1, B2, G1, and G2. With ELISA kits, only total aflatoxins can be tested. With HPLC, one can test a mixture of mycotoxins and determine the efficiency of the adsorbent for each mycotoxin separately.
accurate of these methods. There are positives and negatives to both HPLC and ELISA methods. ELISA kits are easier to use and more cost effective, but they are more prone to matrix effects than HPLC analysis, which can cause false results. Another key limitation to ELISA kits is that they can only reliably quantify in a certain range, e.g. 0.3-10 ppb. Therefore, anything over the limit of the kit (in this example 10 ppb), needs dilutions in order to be accurately calculated, causing additional uncertainty in the results. HPLC does not have this limitation; results can be seen well outside of this range and accurately quantified. Another positive aspect of HPLC is the ability to distinguish between mycotoxins such as aflatoxin B1, B2, G1, and G2. With ELISA kits, only total aflatoxins can be tested. With HPLC, one can test a mixture of mycotoxins and determine the efficiency of the adsorbent for each mycotoxin separately.
Here at Trilogy, we test mycotoxin binders using HPLC methodology. Determinations are performed in triplicate to determine how much of the toxin is bound to the adsorbent. Desorption is then calculated to determine how much of the toxin remains bound as it moves through the animal’s digestive tract. Learn more about mycotoxin binder analysis at Trilogy by visiting our website or viewing our whitepaper on the topic. If interested in setting up a mycotoxin binding study, please contact us.

Share on Social Media: